Featuring insights from prof. Arnaud Bourdin, University of Montpellier
Severe Asthma research is entering a new phase. Over the past decade, biologic therapies have transformed the treatment landscape for patients with uncontrolled disease, particularly those with eosinophilic inflammation. Yet despite these advances, a fundamental challenge remains: many patients still respond unpredictably to treatment. Understanding why some patients benefit while others do not is becoming one of the central questions in severe asthma research, and increasingly, the answer may lie not only in inflammation, but also in the structural changes occurring within the airways and lungs.(1)
Seeing Asthma Through Structural Imaging
Chest CT scans capture detailed information about airway anatomy across the entire lung. However, traditional visual interpretation uses only a fraction of that information and can vary significantly between readers.
Advances in AI-enabled quantitative CT (QCT) analysis are shifting the landscape. By replacing subjective visual interpretation with automated, high-precision measurements, researchers can now evaluate structural airway features with high precision. This quantitative approach enables more accurate assessment of disease extent, the detection of small airway disease, and the identification of early-stage disease pathology. Furthermore, it provides a highly sensitive tool for tracking disease progression and measuring the specific impact of new therapy.
Example of AI-enabled QCT metrics for the assessment of structural lung abnormalities
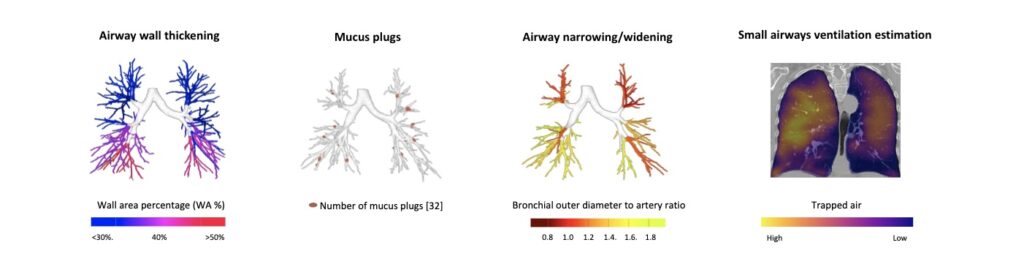
In recent years, these techniques have been applied in multiple research collaborations exploring structural lung disease in conditions such as COPD, cystic fibrosis, bronchiectasis, and severe asthma. In asthma specifically, quantitative CT studies have helped demonstrate how structural abnormalities correlate with disease severity and lung function impairment.(2)
In severe asthma research, several quantitative CT metrics have proven particularly informative. These include measurements of airway wall thickness and lumen dimensions using Bronchus–Artery analysis, mucus plug burden across the bronchial tree, and trapped air reflecting small airway dysfunction(3) derived from paired inspiratory–expiratory CT scans. Studies applying these metrics have demonstrated their ability to objectively characterize structural airway abnormalities and link them to clinical outcomes such as airflow limitation and exacerbation risk in severe asthma.(4,5)
These insights are gradually expanding the way researchers think about asthma, moving beyond purely inflammatory phenotyping toward a more comprehensive view that also includes structural airway characteristics.(2)
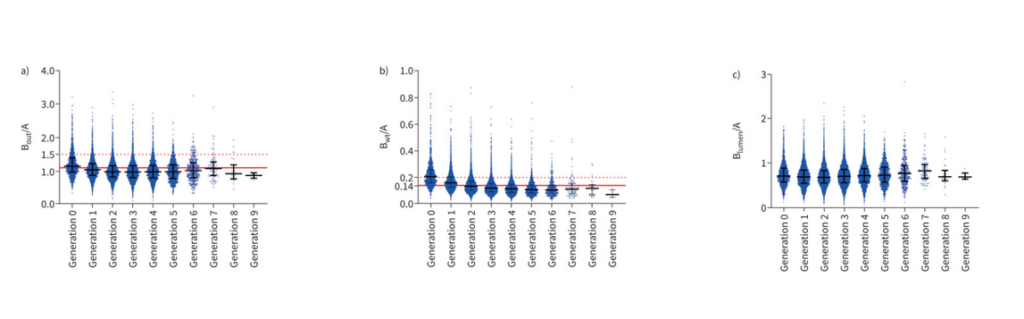
Overview of Bronchus-Artery ratios, quantified with LungQ®BA from CT scans of 131 children with Severe Asthma
A New Question: Do Biologic Therapies Influence Airway Structure?
While biologic therapies are known to reduce eosinophilic inflammation and improve clinical outcomes, an important scientific question has emerged: can these therapies also influence the structural features of airway disease?
Addressing this question requires tools capable of measuring subtle structural changes in the lungs over time, something that has historically been difficult with conventional imaging assessment.
Building on earlier imaging research, collaborations between imaging scientists and leading respiratory centers are beginning to explore how structural imaging can inform treatment response in severe asthma. One of the first studies to investigate this question longitudinally in patients receiving biologic therapy is the BENRALISCAN study NCT03976310, conducted in collaboration with leading respiratory research groups including the University of Montpellier.
The study follows patients with severe eosinophilic asthma and evaluates how airway structure evolves during therapy, including measurements of airway dimensions, mucus plug burden, and air trapping.
As Prof. Arnaud Bourdin, Head of the Department of Respiratory Medicine at the University of Montpellier, explains:
“Small airway disease has long been suspected to play a central role in asthma, but it has historically been difficult to measure non-invasively. Quantitative CT imaging provides a powerful way to capture these structural changes objectively.”
Early findings suggest that while biologic therapy primarily targets inflammation, it may also influence certain structural airway features, including reductions in mucus burden and improvements in airway wall abnormalities.
The results will be presented at the ATS International Conference 2026, marking an important step in understanding how biologic therapies may affect both inflammation and airway structure in severe asthma.
Toward Structural Precision in Asthma
A growing body of imaging research shows that structural airway abnormalities such as mucus plugging, airway remodeling, and small airway dysfunction, can now be measured objectively and reproducibly using quantitative CT analysis, while also providing new evidence of treatment effects by revealing how these structural features respond to therapy.
This opens the door to a new generation of imaging biomarkers that could help researchers better understand treatment mechanisms, monitor therapeutic effects, and ultimately refine patient selection in clinical trials.
In the future, combining inflammatory biomarkers with structural imaging insights may help explain why patients with similar clinical profiles respond differently to therapy and guide more precise treatment strategies.
While further validation studies are needed, the progress made so far suggests that structural imaging is becoming an increasingly valuable component of severe asthma research.
And as these insights continue to evolve, they may help pave the way toward a more precise and individualized approach to treating severe asthma.
Reference publications
- A Tiotiu et al, Biologics and airway remodeling in asthma: early, late, and potential preventive effects, Allergy, 2024 Nov 9 Biologics and airway remodeling in asthma: early, late, and potential preventive effects – PMC
- T Van Der Veer et al, Airway Thickening, Mucus Plugs and Eosinophilia in Bronchiectasis and Asthma, European Respiratory Journal 2024 Airway Thickening, Mucus Plugs and Eosinophilia in Bronchiectasis and Asthma | European Respiratory Society
- H Tiddens et al, Automatic and Manual Quantification of Small Airways Disease on Chest CT of Children with Severe Asthma, American Journal of Respiratory and Critical Care Medicine Automatic and Manual Quantification of Small Airways Disease on Chest CT of Children With Severe Asthma | A69. AN IMAGE’S WORTH: STUDIES IN LUNG IMAGING
- W Van den Bosch et al, Children with severe asthma have substantial structural airway changes on computed tomography, ERJ Open Research 2024 Children with severe asthma have substantial structural airway changes on computed tomography | European Respiratory Society
- W Van den Bosch et al, Automatic detection of mucus plugs on computed tomography scans in severe paediatric asthma, European Respiratory Journal Automatic detection of mucus plugs on computed tomography scans in severe paediatric asthma | European Respiratory Society
